|
Chemists often make general statements about the properties of the elements in a group using descriptive names with historical origins. However, even these small quantities are required for the body to function properly.Īs previously noted, the periodic table is arranged so that elements with similar chemical behaviors are in the same group. These last three metals are not listed explicitly in Table 2.1.2, so they are present in the body in very small quantities. Cobalt is a necessary component of vitamin B-12, a vital nutrient. 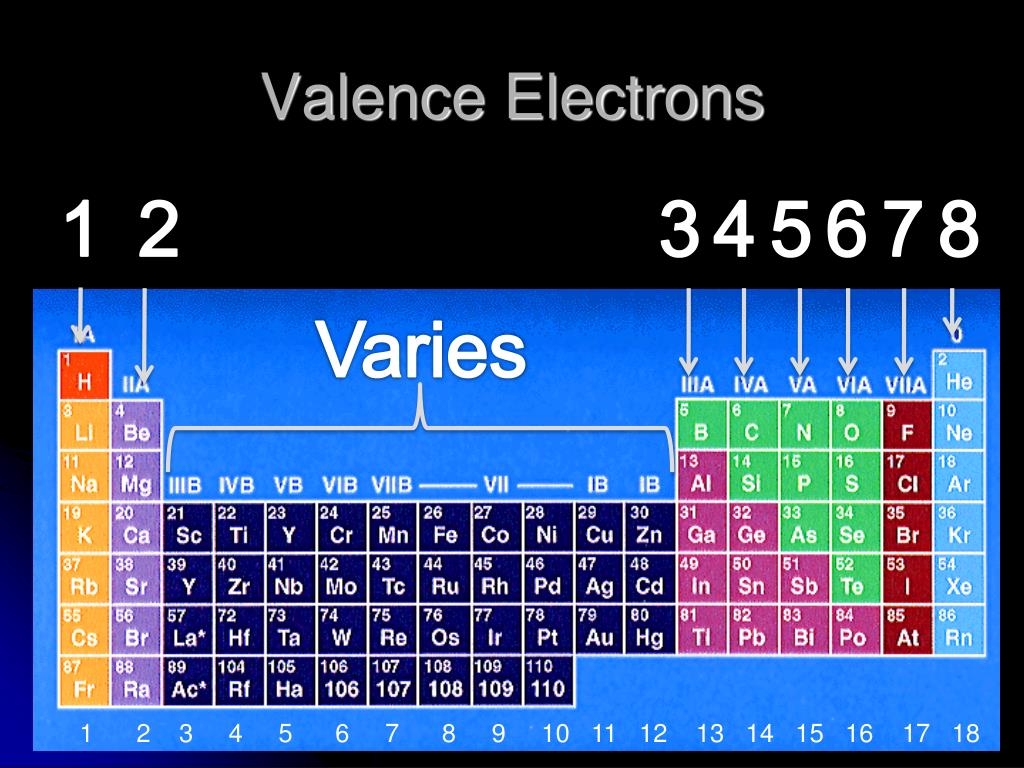
Manganese is needed for the body to metabolize oxygen properly. Copper is also needed for several proteins to function properly in the body. Zinc is needed for the body’s immune system to function properly, as well as for protein synthesis and tissue and cell growth. Other transition metals have important functions in the body, despite being present in low amounts. It is the presence of this particular transition metal in your red blood cells that allows you to use the oxygen you inhale. Each hemoglobin molecule has four iron atoms, which act as binding sites for oxygen. The crucial atom in the hemoglobin protein is iron. Hemoglobin is a relatively large molecule, with a mass of about 65,000 u. Hemoglobin combines with oxygen and carbon dioxide, transporting these gases from one location to another in the body. The critical part of the red blood cell is a protein called hemoglobin. Without red blood cells, animal respiration as we know it would not exist. Red blood cells are cells that transport oxygen from the lungs to cells of the body and then transport carbon dioxide from the cells to the lungs. The chemistry of iron makes it a key component in the proper functioning of red blood cells. Transition metals have interesting chemical properties, partially because some of their electrons are in d subshells. Because iron has relatively massive atoms, it would appear even lower on a list organized in terms of percent by atoms rather than percent by mass. The first element appearing on the list that is not a main group element is iron, at 0.006 percentage by mass. Most of the elemental composition of the human body consists of main group elements. To Your Health: Transition Metals in the Body Each group is located in a different part of the periodic table. Elements are either metals, nonmetals, or semimetals. (2012, December 18) Valence Electrons and the Periodic Table. If the valence shell of an element is full, such as with a noble gas, then the element does not want to gain or lose an electron.įor example, alkali metals, which all have a valency of 1, want to lose that one electron and are likely to form ionic bonds (such as in the case of NaCl, or table salt) with a Group 17 element, which has a valency of 7 and wants to gain that one electron from the alkali metal (Group 1 element) to form a stable valency of 8.įor more on valence electrons and how they're related to the periodic table, I strongly recommend this video:Ĭitations: Tyler Dewitt. They determine how "willing" the elements are to bond with each other to form new compounds. Valence electrons are responsible for the reactivity of an element. You can easily determine the number of valence electrons an atom can have by looking at its Group in the periodic table.įor example, atoms in Groups 1 and 2 have 1 and 2 valence electrons, respectively.Ītoms in Groups 13 and 18 have 3 and 8 valence electrons, respectively. Valence electrons are the electrons present in the outermost shell of an atom. To form a covalent bond, one electron from the halogen and one electron from another atom form a shared pair.įor example, in #"H–F"#, the dash represents a shared pair of valence electrons, one from #"H"# and one from #"F"#. To form an ionic bond, a halogen atom can remove an electron from another atom in order to form an anion (e.g., #"F"^"-", "Cl"^"-"#, etc.). They have one less electron configuration than a noble gas, so they require only one additional valence electron gain an octet. The most reactive nonmetals are the halogens, e.g., #"F"# and #"Cl"#. Nonmetals tend to attract additional valence electrons to form either ionic or covalent bonds. 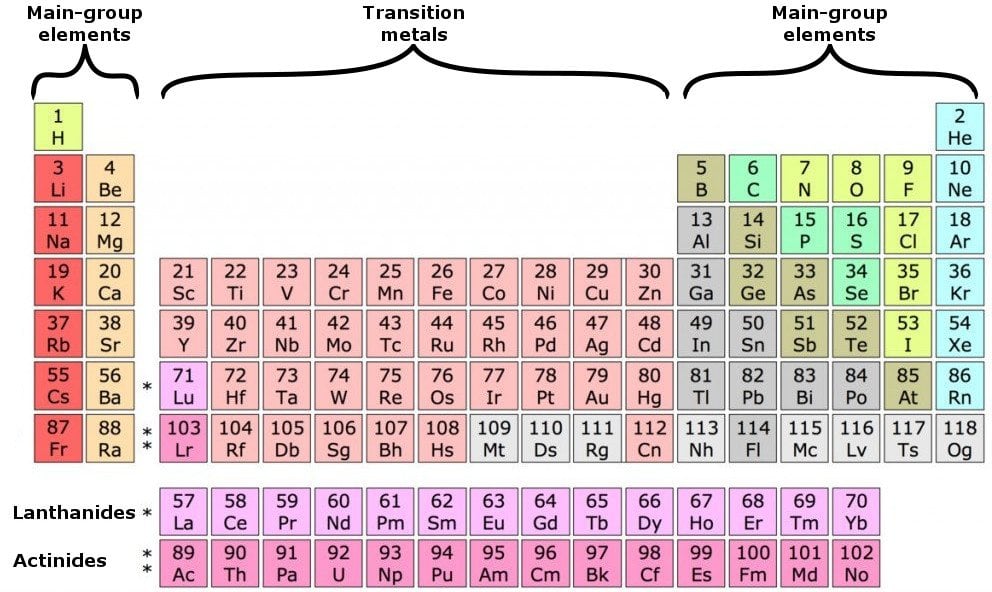
They need to lose only one or two valence electrons to form positive ions with a noble gas configuration. The most reactive metals are those from Groups 1 and 2. 
Generally, elements in Groups 1, 2, and 13 to 17 tend to react to form a closed shell with a noble gas electron configuration ending in #ns^2 np^6#. Elements whose atoms have the same number of valence electrons are grouped together in the Periodic Table.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
